Your Cart is Empty
FREE UK DELIVERY for all Vikan Orders over £150 GBP. MOQ £70.
It is an unstable gas meaning that it readily reacts with other substances or spontaneously breaks down. The half life of Ozone gas is often quoted as 20 minutes but will depend on the environment and media it is present in.
Ozone is produced naturally and is present in varying concentrations throughout the Earth’s atmosphere. In the regions and natural concentrations that Ozone occurs in our atmosphere, it not only protects life from the harmful effects of solar ultraviolet radiation but is also very effective at destroying bacteria, viruses and odours.
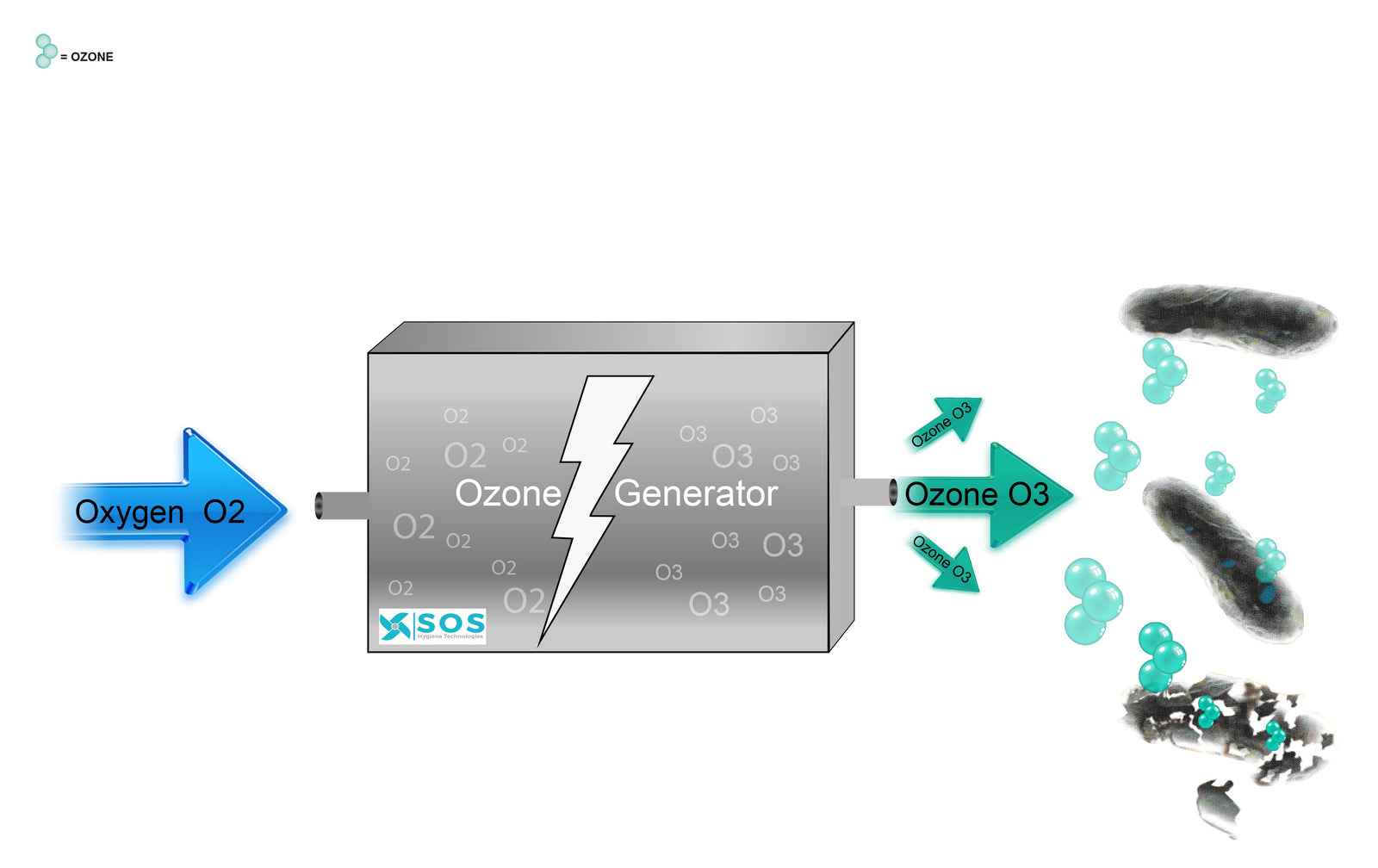
Ozone is created from air and is a highly reactive gas which is produced when the oxygen in the air interacts with either electricity or UV light. Ozone forms when atmospheric electrical discharges (Thunderstorms/Lightning) or highly energetic solar radiation strikes molecules of Oxygen (O2) this causes the Oxygen molecules to split apart. If a freed atom then collides with another O2 molecule it may combine to form Ozone (O₃). Ozone is also known as “activated” oxygen and is an extremely powerful and effective natural cleansing agent.
Ozone is the second most powerful oxidant known. Ozone is an unstable gas it decays readily especially when it comes into contact with organic compounds. It then partly reverts to O2 and at the same time releases a single highly reactive O atom. It then partly reverts to O2 and at the same time releases a single highly reactive O atom. When the highly reactive O atom is released and is in contact with a bacteria or virus this single atom attacks the cell membrane. Initially Ozone disrupts the glycoproteins, glycolipids and certain amino acids such as tryptophan in the bacterial membrane. It also acts upon the sulfhydrl groups and some enzymes resulting in the disruption of normal cellular activity. Bacterial death is rapid and is often attributed to changes in the cell permeability followed by cell lysis.
When Ozone reacts with Volatile Organic Compounds (VOCs) such as odours the free oxygen atom reacts with the VOCs oxidising them to create harmless or non odorous substances that are predominantly CO2 and H2O.
• Destroys bacteria on food and protects at all stages from farm to fork.
• Ozone is effective on all orientations and destroys pathogens in areas where traditional disinfectants are unable to reach.
• Ozone is effective in dry (Gas phase) and wet (Aqueous phase) environments.
• Ozone is not subject to Maximum Residue Levels (MRLs).
• Ozone reduces the need for harsh chemicals, the handling and storage of chemicals and removes the consumable cost of chemicals.
• Ozone can help reduce product fails and recalls, which mitigates the risk of damage to the company brand and reputation.
Formula:0₃
Density:2.14kg/mᵌ
Molar Mass:48g/mol
Boiling point:-112˚C
Melting Point:-192˚ C
Odour:Bleach/Chlorine smell at concentrations above 0.03ppm (v/v)
Other Info:Ozone Gas is heavier than air. Ozone can be dissolved into water to be used as a disinfectant
Artificially generated ozone is just as effective as that naturally occurring. It can be generated in very high concentrations but should be carefully controlled. Here at SOS Hygiene Technologies Limited we generate ozone using Primozone Cold Plasma technology – dielectric barrier discharge.